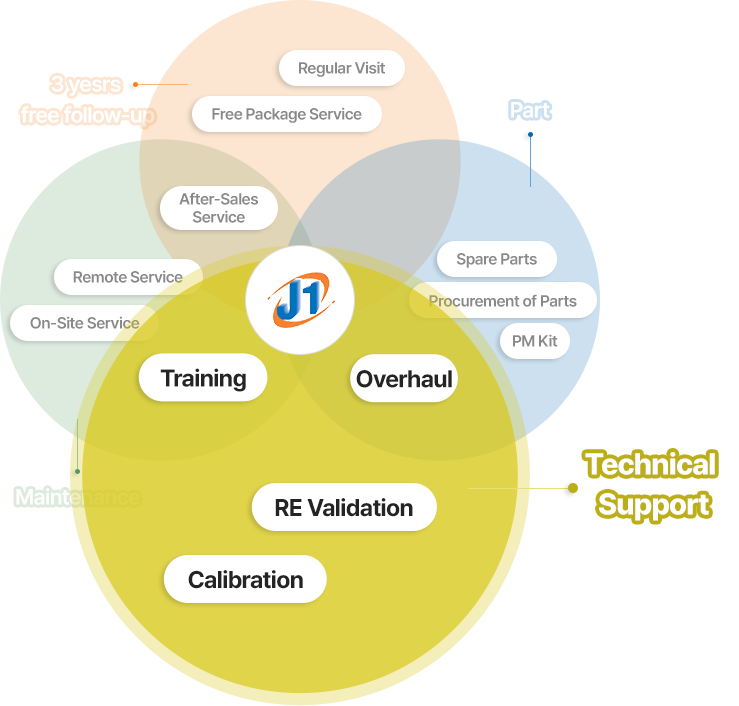
Our Service
Technical Support
Providing Training for High-Quality Pharmaceuticals
Technical Support
Training Service
J1 TnM's training service provides education using verified global manufacturers' programs and
expert technicians' know-how, ensuring optimal equipment efficiency in the production field.
Customized Training Course
We provide customized training tailored to the production environment and trainees. Through the delivery of fundamental equipment information and knowledge, we ensure users become familiar with operating the equipment efficiently.
Product Manufacturing & Maintenance Training
We provide expertise on preventing failures, reducing errors, and improving production efficiency, enabling longer and more effective use of the equipment.
Equipment Setup & Software Training
We provide training from essential initial equipment setup to the interface operation required for actual production and validation.
This ensures the collection, recording, and management of data, allowing customers to analyze the data in the future.
Calibration
Periodic calibration is essential for the smooth operation of equipment. This enhances production quality stability and ensures reliability.
J1 engineers, who have completed professional training from the manufacturer, perform calibration using official measuring instruments from the manufacturer.
Ensuring Stability
For sensors, missing the calibration schedule may compromise the uniformity of the produced products. For reference, the recommended calibration interval for Kilian tablet presses is one year.
Cost Reduction
J1 has engineers qualified to perform Kilian tablet press calibration directly.
Regulatory Compliance
Pharmaceutical machinery must comply with strict guidelines and safety standards Regular calibration helps adhere to regulations set by health authorities cGMP compliance guarantee, meeting BSI (British Standards Institution) standards, implementation of safety functions, etc.
Re-Validation
If the equipment has been relocated or upgraded, you can receive support for re-validation
according to the manufacturer's optimized documentation format and procedures.
Cost Optimization
If additional validation is required, such as equipment relocation, support is available. At the same time, inspections can be conducted based on the extensive knowledge and experience of equipment experts. Immediate action is also possible.
Global Standard
IQ, OQ, etc. can be re-executed according to global standard formats..
Regulatory Compliance
Pharmaceutical machinery must comply with strict guidelines and safety standards Re-Validation helps adhere to regulations set by health authorities cGMP compliance guarantee, meeting BSI (British Standards Institution) standards, implementation of safety functions, etc.
Overhaul
J1 TnM's overhaul restores reliable high-quality standards to machinery, giving new life to the core components of existing equipment.
Upgrade your equipment at a lower cost than purchasing new machinery. Not only hardware but also software can be upgraded to the latest version.
Additionally, any issues discovered during the engineer’s overhaul period can be addressed immediately.
Cost Efficiency
Investing in new machinery can be costly. Overhauling existing equipment provides a cost-effective alternative while achieving similar or even better performance.
Quality Assurance
Overhaul ensures that the machine continues to produce high-quality products. You can use the latest version of the equipment at a lower cost.
Regulatory Compliance
Regular overhauls of older equipment ensure compliance with new regulations set by health authorities. cGMP compliance guarantee, meeting BSI (British Standards Institution) standards, implementation of safety functions, etc.






